Cardinal Health MarketSM
The legacy Cardinal.com Medical Ordering site has been replaced with Cardinal Health MarketSM, a new product experience designed with you in mind.
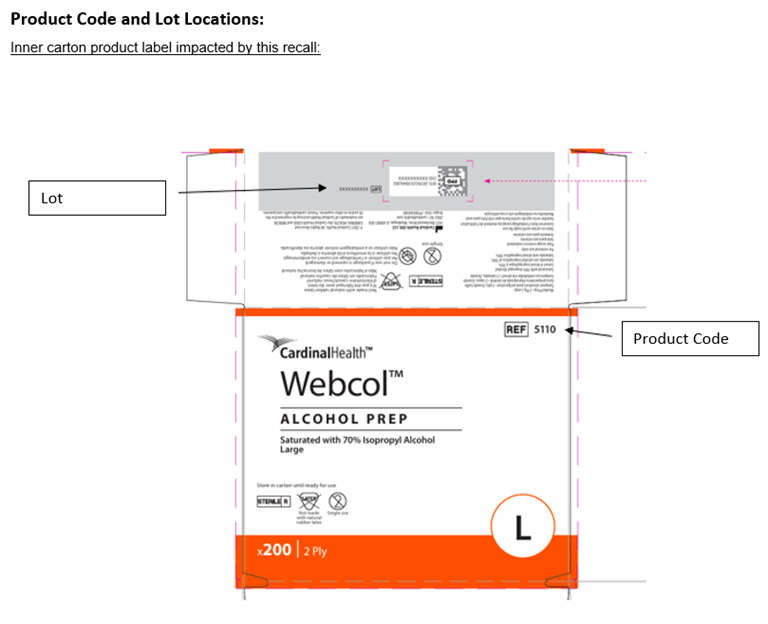
DUBLIN, Ohio, March 19, 2026 – Cardinal Health has issued a voluntary recall for select lots of Webcol™ Large Alcohol Prep Pads (70% isopropyl alcohol) to the consumer level. The product is being recalled due to microbial contamination identified as Paenibacillus phoenicis.
There is a reasonable probability that use of the contaminated pads may result in local and systemic infections in at-risk populations; patients with impaired immune response, including patients with cancer receiving chemotherapy or poorly controlled diabetes, may develop life-threatening infections such as bacteremia or central nervous system infections.
The Webcol™ Alcohol Prep Pad is used for external use only and utilized for cleansing.
The product was distributed in the U.S., Puerto Rico and Japan between September 2025 and February 2026.
Affected Webcol™ Alcohol Prep Pad lots include the following lot numbers:
|
25H065362 |
25J013962 |
25J034762 |
25J064562 |
25K004962 |
25K041262 |
25K065062 |
25L015362 |
|
25H065462 |
25J014062 |
25J035762 |
25J064762 |
25K005062 |
25K041362 |
25K065162 |
25L015462 |
|
25H066062 |
25J014862 |
25J035862 |
25J064862 |
25K006162 |
25K041462 |
25K065262 |
25L015662 |
|
25H066162 |
25J014962 |
25J035962 |
25J064962 |
25K006262 |
25K041562 |
25K065362 |
25L016362 |
|
25H066262 |
25J033962 |
25J050362 |
25J065762 |
25K006362 |
25K041662 |
25K064562 |
25L015962 |
|
25H066362 |
25J034862 |
25J050462 |
25J066262 |
25K005162 |
25K040462 |
25K064662 |
25L016062 |
|
25H066462 |
25J034062 |
25J050562 |
25J066362 |
25K006462 |
25K040562 |
25K065462 |
25L030162 |
|
25J013462 |
25J034162 |
25J050962 |
25K004062 |
25K019662 |
25K040662 |
25K065562 |
25L058062 |
|
25J013562 |
25J034262 |
25J051062 |
25K004162 |
25K020862 |
25K040762 |
25K065662 |
25M022362 |
|
25J014362 |
25J034362 |
25J051262 |
25K005262 |
25K020962 |
25K040962 |
25K066262 |
25M028462 |
|
25J014462 |
25J034462 |
25J051362 |
25J065962 |
25K019762 |
25K041062 |
25K066162 |
|
|
25H065962 |
25J034962 |
25J051462 |
25J065062 |
25K019862 |
25K041162 |
25K064862 |
|
|
25H065762 |
25J035062 |
25J051762 |
25J065162 |
25K021062 |
25K041762 |
25K065862 |
|
|
25H065862 |
25J035162 |
25J051862 |
25J066062 |
25K021162 |
25K041862 |
25K079962 |
|
|
25H066862 |
25J035262 |
25J051962 |
25J066162 |
25K021262 |
25K041962 |
25K080962 |
|
|
25H066962 |
25J035362 |
25J075762 |
25K004262 |
25K019962 |
25K042162 |
25K081062 |
|
|
25J013162 |
25J013662 |
25J051162 |
25K004362 |
25K020062 |
25K042262 |
25K080062 |
|
|
25J013262 |
25J014562 |
25J052062 |
25K004462 |
25K021362 |
25K040862 |
25K080162 |
|
|
25J013362 |
25J014662 |
25J052162 |
25K004562 |
25K021462 |
25K042062 |
25K080262 |
|
|
25J014162 |
25J014762 |
25J052262 |
25K004662 |
25K021562 |
25K063862 |
25K080362 |
|
|
25J014262 |
25J034562 |
25J052362 |
25K005362 |
25K020162 |
25K020562 |
25K080462 |
|
|
25H065562 |
25J034662 |
25J064362 |
25K005462 |
25K020262 |
25K020362 |
25K081162 |
|
|
25H065662 |
25J035462 |
25J064462 |
25K005562 |
25K020662 |
25K020462 |
25K081262 |
|
|
25H066562 |
25J035562 |
25J064662 |
25K005662 |
25K021662 |
25K021962 |
25K081362 |
|
|
25H066662 |
25J035662 |
25J065262 |
25K005762 |
25K021762 |
25K022062 |
25K081462 |
|
|
25H066762 |
25J050662 |
25J065362 |
25K004762 |
25K021862 |
25K063962 |
25K081562 |
|
|
25J012962 |
25J050762 |
25J065462 |
25K004862 |
25K040062 |
25K064062 |
25K080562 |
|
|
25J013062 |
25J050862 |
25J065562 |
25K005862 |
25K040162 |
25K064162 |
25L014062 |
|
|
25J013762 |
25J051562 |
25J065662 |
25K005962 |
25K040262 |
25K064262 |
25L014162 |
|
|
25J013862 |
25J051662 |
25J065862 |
25K006062 |
25K040362 |
25K064462 |
25L015262 |
Cardinal Health notified its customers by overnight mail on March 2, 2026, with instructions to:
Consumers with questions regarding this recall can contact Cardinal Health at GMB-FieldCorrectiveAction@cardinalhealth.com or call 800-292-9332 Monday-Friday between 8am and 5pm EST.
Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.